Minimally Invasive Back Surgery Gives Young Mom Her Life Back
March 6, 2024
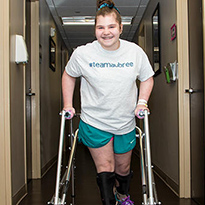
Maricela Cedillo began experiencing minor back flare-ups in her mid-twenties and early thirties, which she attributed to physical activities like running and working out. “My back would hurt,” she says, “but the pain would go away within a day or two.”